From Reuters.com…
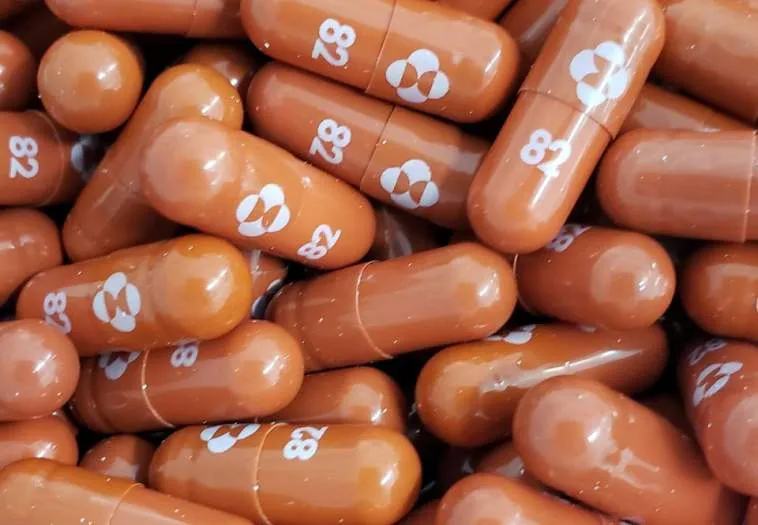
A pill developed by U.S. drugmaker Merck could half the chances of dying or being hospitalized for those most at risk of contracting severe COVID-19, with experts hailing it as a potential breakthrough in how the virus is treated.
If it gets authorization, molnupiravir, which is designed to introduce errors into the genetic code of the virus, would be the first oral antiviral medication for COVID-19.
Merck (MRK.N) and its partner Ridgeback Biotherapeutics plan to seek U.S. emergency use authorization for the pill as soon as possible and to make regulatory applications worldwide.
“This is going to change the dialogue around how to manage COVID-19,” Merck Chief Executive Robert Davis told Reuters.